![PDF] Anomalies in isothermal compressibility and exponent of pressure in spin-orbit-coupled degenerate Fermi gases | Semantic Scholar PDF] Anomalies in isothermal compressibility and exponent of pressure in spin-orbit-coupled degenerate Fermi gases | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c7385b47eef5baa94554ca8fef1d7d1d2db20349/1-Figure1-1.png)
PDF] Anomalies in isothermal compressibility and exponent of pressure in spin-orbit-coupled degenerate Fermi gases | Semantic Scholar
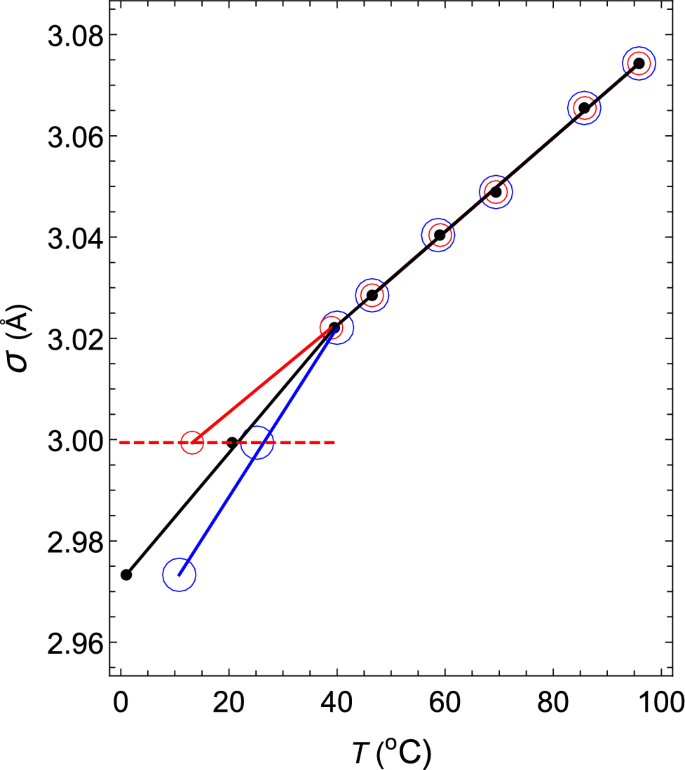
Thermodynamic mechanism of the density and compressibility anomalies of water in the range − 30 < T (°C) < 100 | Scientific Reports
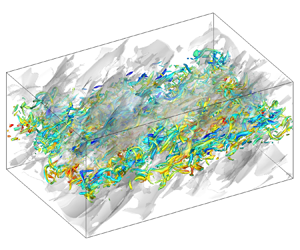
Compressibility effects on statistics and coherent structures of compressible turbulent mixing layers | Journal of Fluid Mechanics | Cambridge Core

SOLVED:Generally, volume expansivity βand isothermal compressibility κdepend on T and P. Prove that: ((∂β)/(∂P))T=-((∂κ)/(∂T))P
![Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram](https://www.researchgate.net/publication/329439239/figure/fig3/AS:961808689008649@1606324469311/Calculated-isothermal-compressibility-kTdocumentclass12ptminimal.png)
Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram

SOLVED: Isothermal compressibility, Kr (kappa defined as: Derive the formula that - van der waals gas would have for kappa Show the unit analysis on your answer in part and check if

Density, Speed of Sound, Compressibility and Related Excess Properties of Methane + n-Heptane at T = 303.15 K and p = 10 to 70 MPa | SpringerLink

SOLVED:The isothermal compressibility κof a gas is defined in Problem 1.17, and its value for an ideal gas is shown to be 1 / P Use implicit differentiation of V with respect
![Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram](https://www.researchgate.net/publication/329439239/figure/fig3/AS:961808689008649@1606324469311/Calculated-isothermal-compressibility-kTdocumentclass12ptminimal_Q640.jpg)
Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram

The compressibility kappa of a substance is defined as the fractional change in volume of that substance for a given change in pressure : kappa = - 1V dVdP (a) Explain why
![SOLVED: [6 marks] This problem gives practice with taking derivatives! The isothermal compressibility Kr (kappa) describes how the volume of a substance changes with pressure at constant temperature. Given Page of 3 SOLVED: [6 marks] This problem gives practice with taking derivatives! The isothermal compressibility Kr (kappa) describes how the volume of a substance changes with pressure at constant temperature. Given Page of 3](https://cdn.numerade.com/ask_images/8fd0309efe92472f8d87f1bd77df5223.jpg)
SOLVED: [6 marks] This problem gives practice with taking derivatives! The isothermal compressibility Kr (kappa) describes how the volume of a substance changes with pressure at constant temperature. Given Page of 3
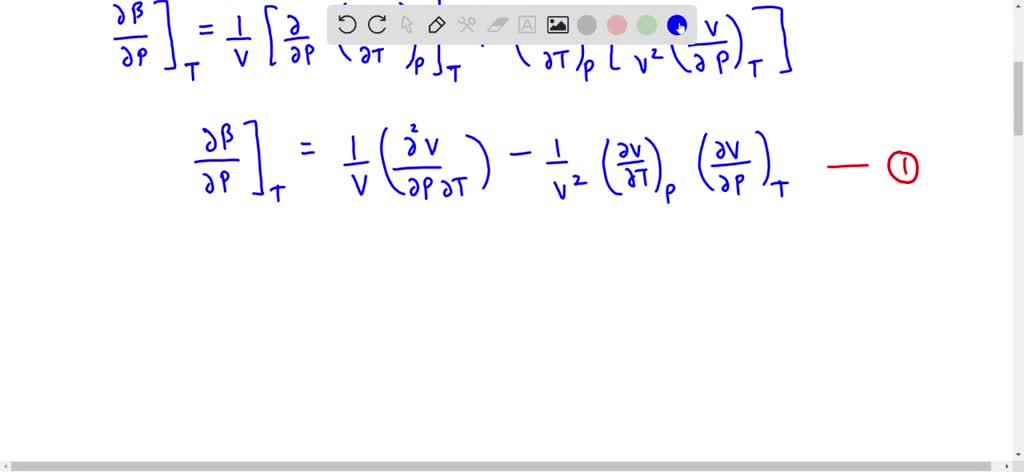





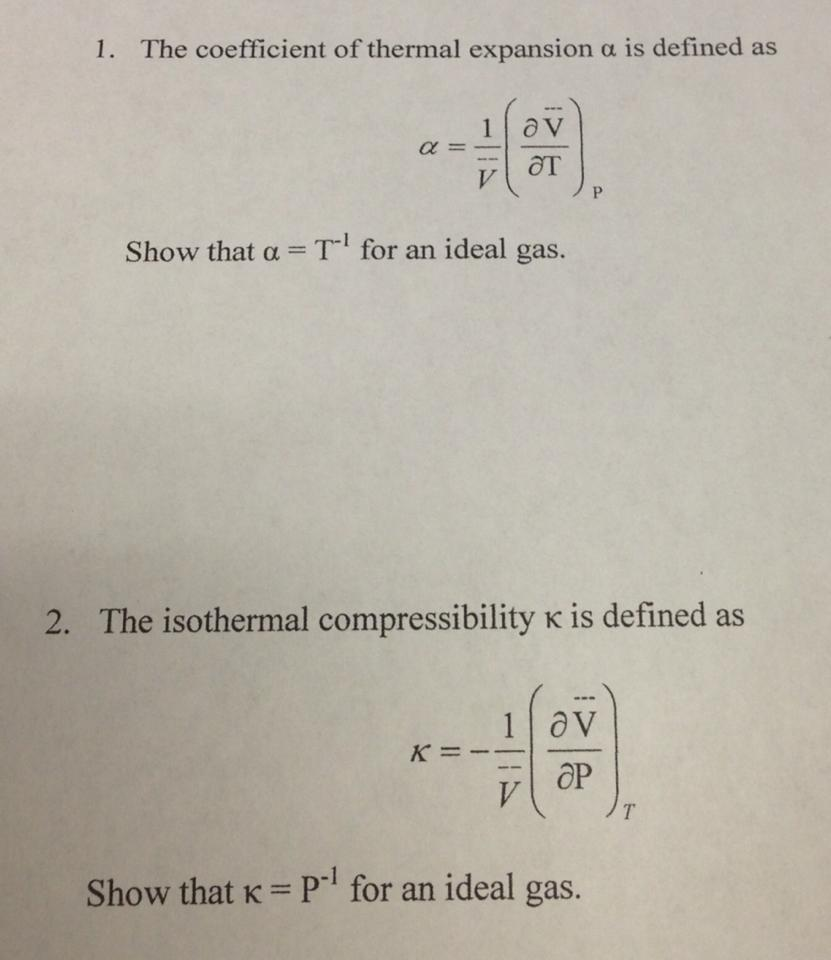





![Isothermal compressibility (κT\documentclass[12pt]{minimal}... | Download Scientific Diagram Isothermal compressibility (κT\documentclass[12pt]{minimal}... | Download Scientific Diagram](https://www.researchgate.net/publication/355714799/figure/fig8/AS:1084113842896899@1635484290073/Isothermal-compressibility-kTdocumentclass12ptminimal-usepackageamsmath.png)